
March 2026
Disruptors and Trends for 2026
- 2026 has the potential to be an exciting year for oncology drug approvals – with nearly 20 drugs pending review and a few high-profile drugs nearing pivotal read out; we conducted a poll recently that found clear enthusiasm for daraxonrasib’s disruptive potential – as the first pan-RAS inhibitor.
- Aside from the disruption potential of these agents, key trends impacting the work we are doing include the implications of novel treatments moving into the early-stage setting, use of ctDNA/MRD, and the wave of novel modalities winding their way through clinical testing.
Targeted therapy has been the driver of new oncology approvals in recent history, but a more diverse mix of MOAs have been approved in the past few years
New Oncology Agents – 2021 to Present
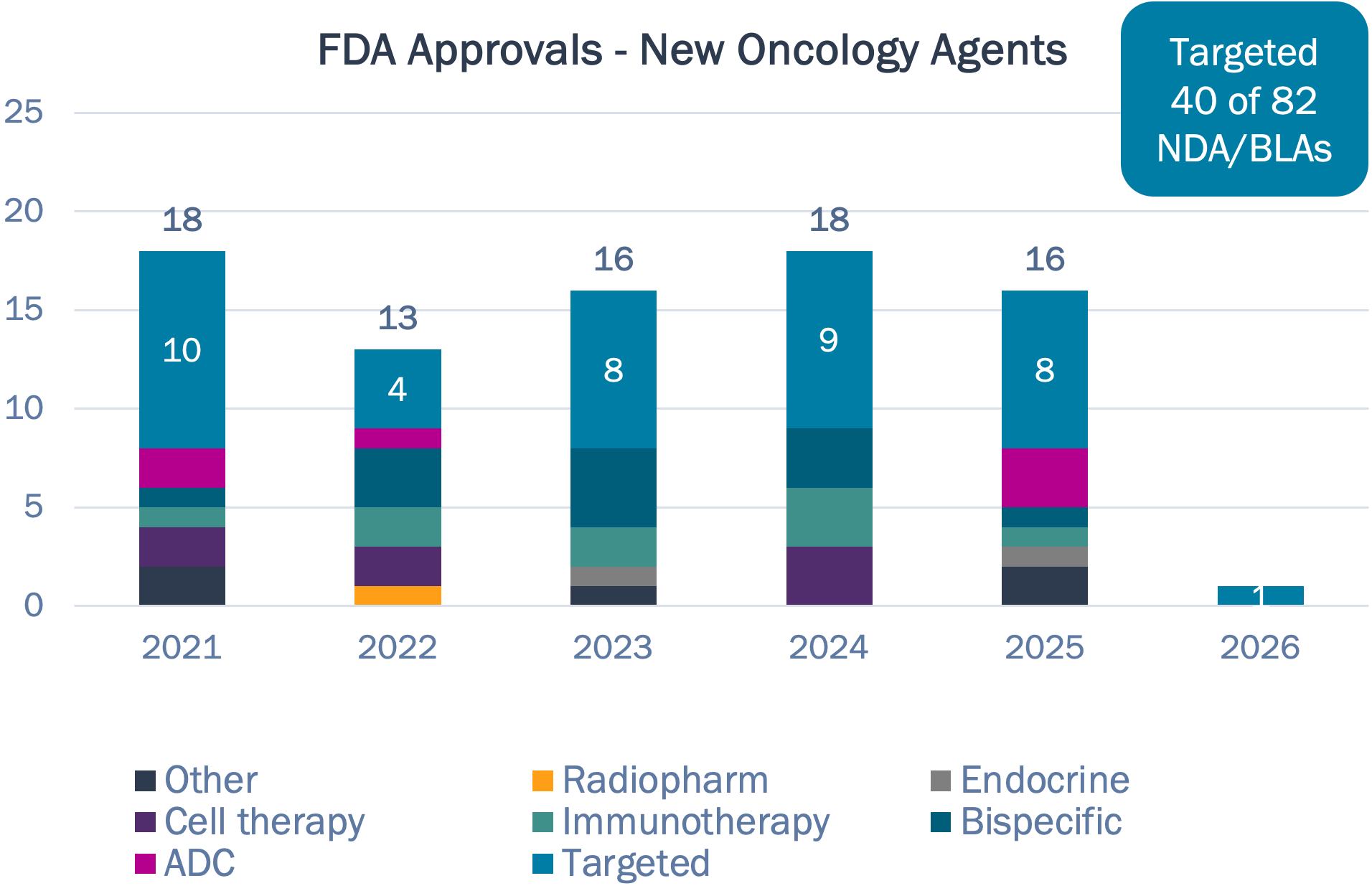
Targeted therapies account for <50% of new approvals in past 5 years, down from >65% in the prior 5 years
Half of drugs approved in 2024 had novel targets, while 2025 only had 3
The first cell therapies and T-cell engager for solid tumors approved in 2024
Wave of ADC dealmaking yet to crest in clinical practice — Datroway (TROP2) and Emrelis (MET) first new ADCs approved in 3 years
Source: FDA; Bluestar analysis
Among the most exciting potential filings/approvals for 2026 are Roche’s next gen G12Ci, divarasib, RevMed’s pan-RAS, daraxonrasib, and Summit’s PD-1xVEGF, ivonescimab
2026 New Oncology Agents
| Targeted | Cell/Gene Therapy |
IO | Bispecific | ADC | Endocrine | Radiopharm/Other | |
|---|---|---|---|---|---|---|---|
| Approved |
Lifyorli (GR) |
||||||
| Filed |
Gedatolisib (PI3K) Sonrotoclax (bcl-2) Zidesamtinib (ROS1) Zipalertinib (EGFRex20) Zanzalintinib (multi-TKI) Bezaclustinib (KIT) Rusfertide (hepcidin) |
Tabelecleucel (EBV) Orca-T (T-cells) Anito-cel (BCMA) |
RP-1 (oncolytic virus) Sasanlimab (PD-1) Cretostimogene |
Odronextamab Ivonescimab |
Pivekimab sunirine |
Vepdegestrant Camizestrant Giredestrant |
177Lu-DOTATOC Pegargiminase |
| Filing Expected |
Iberdomide (CELMoD) Ozekibart (DR5) Mezigodomide |
||||||
| Pivotal Readout Expected in 2026 |
Divarasib (G12C) Daraxonrasib Mevrometostat (EZH2) |
Fianlimab (LAG-3) Galinpepimut-S Gotistobart (CTLA4) |
SSGJ-707 |
Sonesitatug vedotin Sigvotatug vedotin Trastuzumab |
Above: Bold – novel target; Crossout – negative trial or filing withdrawn
Drugs to watch in 2026:
- Roche’s divarasib (G12Ci) represents a potential 2nd generation, going head-to-head against approved sotorasib and adagrasib in 2L NSCLC.
- RevMed’s daraxonrasib (panRAS) could change 2L PDAC, cracking open the KRAS market beyond G12C inhibitors.
- Ivonescimab (Summit, PD-1xVEGF) filed for 2L+ EGFRm NSCLC — will FDA approve despite lack of OS benefit at final analysis?
Source: TrialTrove; FDA; Company financial reports; Bluestar analysis
In a Bluestar poll, most selected daraxonrasib as the pivotal-stage drug with greatest potential disruption potential; there is more uncertainty around each of the other poll options
Bluestar Poll Results
Which Potential 2026 Approval or Pivotal Read Out Will Have the Most Impact on the Oncology Treatment Landscape?
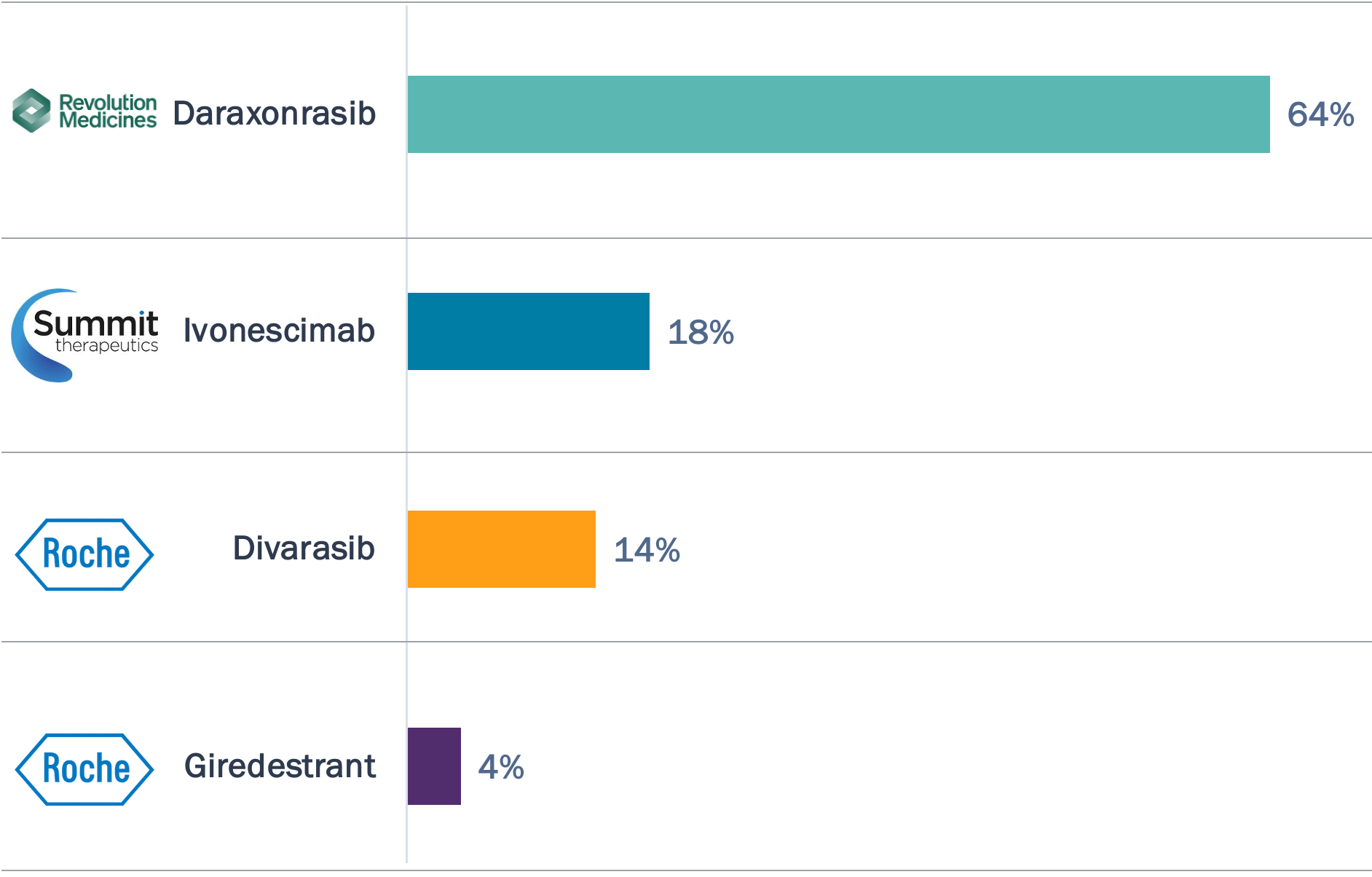
- Daraxonrasib garners the majority of votes, with its compelling 8.5 mos mPFS and 13.1 mos mOS in 2L G12X PDAC; there are also ongoing pivotal trials in adjuvant and 1L PDAC and 2L NSCLC.
- Ivonescimab’s initial FDA filing is uncertain given the lack of OS benefit in the HARMONi trial, but global Ph3 read outs in 1L non-AGA NSCLC and 1L CRC expected in 2028.
- Divarasib is aiming to demonstrate superiority to approved KRAS G12Ci—as well as move earlier in the treatment paradigm, with an open Ph3 trial in 1L NSCLC.
- Giredestrant has two positive Ph3 trials in HR+/HER2- breast cancer — evERA (2L+) and lidERA (adjuvant) — but recent failure of persevERA (1L) raises questions about lidERA ‘all comer’ signal.
Source: TrialTrove; FDA; Company financial reports; Bluestar analysis
Key Trends and Questions for 2026
Key Trends...
...Emerging Questions
Novel treatment modalities (IO, targeted therapies, ADCs,) are securing approval in the early-stage setting
How will these therapies impact the metastatic drug-treated population? Will recurrent patients be eligible for retreatment or need novel options (pushing up 2L)?
Selecting the ‘right’ patient and right duration: using ctDNA/MRD to determine treatment
Are surrogates like ctDNA and MRD ready for prime time in liquid and solid tumors? Should they be used to inform treatment escalation or de-escalation?
Same target - different modality: defining the optimal positioning and sequencing for ADCs, T-cell engagers, radioligands, cell therapies
Which approach is best – by tumor, setting, patient characteristics? What drives resistance - Can the same target be used across lines of therapy?
The next wave of innovation: bispecifics (two targets, or three!, are better than one), in vivo CART, novel ADC payloads, and degraders
Is the next wave of truly innovative molecules on the way? How can these new approaches address unmet need?